Research
Publications | Thesis / Dissertations | Mississippi Agricultural and Forestry Experiment Station
Research progress in the Department of Biochemistry, Molecular Biology, Entomology and Plant Pathology is focused on topics to advance agriculture and public health in Mississippi, the southern region and the nation. The departmental faculty addresses a wide-range of research topics ranging from pest and plant interactions, human diseases and public health, commodity improvement (e.g., rice, cotton, corn, biofuels, etc.), taxonomy and systematic biology, environmental toxicology, molecular biology and pharmacology.
Biochemistry and Molecular Biology
Advancing Amphibian Populations Through Reproductive Research

Scientists with the MSU Department of Biochemistry, Molecular Biology, Entomology and Plant Pathology are attempting to increase populations of a number of vulnerable amphibian species held in captive breeding programs, specifically gopher frogs. Unfortunately, encouraging frogs to mate naturally in captivity is not always easy; however, assisted reproductive technologies, such as exogenous hormone administration, are being used to bypass these cues and induce the production and release of sperm and eggs for in vitro fertilization. The ability to collect gametes in this way also allows researchers the opportunity to investigate protocols for the long-term frozen storage of sperm and embryos through cryopreservation.
Developing Reproductive Technologies for Salamanders

Scientists at MSU, in partnership with the Memphis Zoo, are studying model amphibious species like the tiger salamander to improve the reproduction protocols of the endangered salamander genus. These protocols will initially be applied to four targeted, endangered salamander species native to the United States. When successful, this research could lead to the breeding and release of captive born salamanders to the wild in order to bolster threatened populations.
Luminating Infections in Pregnant Mares
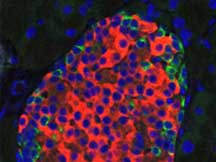
Infections are the leading cause of abortion, stillbirth and preterm delivery in mares. MAFES scientists have developed a new approach to understanding the infection process in pregnant mares by using biophotonic imaging and modified bacteria with luminescent characteristics. In other words, the technique allows researchers to capture real-time pictures of glowing bacteria as they spread through a mare's body. The method allows scientists to track pathogens in a minimally invasive procedure.
MSU Professor Finds Pandas May Aid Biofuel Production

MAFES scientists are looking to microbes in panda excrement that breakdown woody materials as a possible means to biofuel production. These microbes might help overcome one of the major challenges to producing biofuels: breaking down the raw plant materials used to make the fuels. The findings have garnered national attention as the reproduction of these microbes could contribute to developing alternative fuels that don't interfere with food crops and could also save a great deal of money.
Read More
MSU Researcher's Study Leads to Swamps, 'Gators'

A Mississippi State University biologist's fascination with crocodiles has brought together researchers from the United States and Australia to study the genetic building blocks of a reptile order. In the process, they hope to discover ways to conserve endangered animals, harness the antibiotic properties of alligator blood and isolate the genes that determine gender. Such significant genetic information could have a positive impact on wildlife conservation.
Read More
Open Spaces, Endless Possibilities

Dr. JoVonn Hill, a MAFES scientist and assistant professor in the Department of Biochemistry, Molecular Biology, Entomology and Plant Pathology, has discovered a little over 30 new grasshopper species. Hill is part of a consortium of academic, federal, and state biologists brought together by Southeastern Grassland Initiative, or SGI, who are all invested in finding ways to save grassland ecosystems across the Southeast. The group recently published a white paper for the United States Geological Survey, that provides recommendations on improving species assessments under the U.S. Endangered Species Act. The team recommended that the U.S. Fish and Wildlife Service (USFWS) adopt an ecosystem approach to species conservation in southeastern grasslands, noting that such an approach would prove beneficial for both cost-savings and conservation alike. MSU's partnership with SGI is growing as the consortium continues to collaborate on several projects aimed at saving southeastern grasslands.
Read More
Snap, Crackle, Pop! Conversations in a Rice Cell

Dr. Zhaohua Peng and his research group are studying the molecular "conversations" that occur between a rice cell wall and its nucleus. Dr. Peng and his research group have shown that removal of a rice cell wall results in substantial changes in the organization of the chromatin within the nucleus. The communication that occurs between the nucleus and cell wall is likely the result of interactions between various macromolecules, and Dr. Peng's team is currently working to identify these macromolecules and describe how they interact with one another.
Targeting Tomatoes

From herbicide tolerance to gene modification, tomatoes are being studied to help farmers grow the popular fruit with fewer losses or injuries to the plants. Dr. Sorina Popescu, assistant professor in biochemistry, molecular biology, entomology and plant pathology, is working to understand how tomato plants respond to pathogens at the molecular level, and editing the plants for genetic susceptibility using CRISPR (clustered regularly interspaced short palindromic repeats) technology. The pathogen Popescu is studying is called Pseudomonas syringae and causes stunting and yield loss, particularly if young plants are infected. Most pathogens have effectors that act in very similar ways, and Popescu believes the findings on tomato plants can be extended to other vegetable crops to make them less susceptible to pathogens.
Read More
The Fingerprint of Disease

Drs. Florencia Meyer, associate professor, Dr. Carrie Vance, associate research professor, both in the Department of Biochemistry, Molecular Biology, Entomology, and Plant Pathology and scientists in the Mississippi Agricultural and Forestry Experiment Station, have teamed up with Dr. Amelia Woolums, professor in the Department of Pathobiology and Population Medicine in the College of Veterinary Medicine, to research ways to better diagnose bovine respiratory disease (BRD). BRD is the most significant disease impacting the American feedlot industry and expands as a global problem. The team used Near Infrared Spectroscopy (NIRS) to find a better way to diagnose BRD. The current protocol is to prophylactically treat animals exhibiting any BRD symptoms, whether they have the disease or not. By having a NIRS device that tests animals quickly and accurately, this would significantly reduce the amount of antibiotics that are used on animals that do not need them. So, researchers are developing a diagnostic strategy for the early detection and treatment of animals developing BRD. This will be a means to analyze a global set of health parameters in a rapid assay that can determine the specific health status of animals at risk of developing BRD. Modeling what the disease profile looks like makes identifying BRD more like a fingerprint than a single assay. A significant percent of cattle goes unidentified and untreated with BRD, which is why better diagnostic tools need to be developed. The current model the team has created has an overall accuracy of 83.3%, with a sensitivity of 79.2%, and a specificity of 87.5%, which is much higher than the current BRD diagnosis.
Read More
Toxin-free Castor Would Be Major Help to Industry

To make toxic castor beans—the origin of castor oil—a commercially viable U.S. crop, MAFES scientists are trying to discover a way to genetically modify the plant so that either the gene that produces the toxin is no longer expressed or the toxin is no longer produced.
Read More
Entomology
Ant Study at Noxubee Refuge Suggests Healthy Ecosystem

It is no secret that many ants live beneath the leaves, bark and soil of the Noxubee National Wildlife Refuge, but no one knew how diverse the population was until Mississippi State University entomologists dug up the facts. Their findings show that imported fire ants and other exotics have not displaced the natives. More importantly, the distribution and diversity of ants at the refuge indicate a well-managed, healthy ecosystem.
Read More
Bye-Bye Bay Leaf

While ambrosia beetles are believed to have been introduced into the U.S. from Asia in the early 2000s, they were not known to cause damage to healthy trees until the Laurel wilt epidemic. Laurel wilt can readily kill any plant in the family Lauraceae, which is more than a dozen species including swampbay, avocado, sassafras, and more. If Laurel wilt continues at its present pace, consumers might have to say goodbye to bay leaf, produced by the swampbay; guacamole, produced by avocado trees; and spicy gumbo, which contains sassafras. MAFES researchers are steadily working to understand both the disease and the beetles that carry it, having already uncovered that the beetles carrying Laurel wilt are all genetically identical. They have also produced a spatial model that revised the prediction of the disease's spread in hopes of finding a solution to save the vulnerable trees in the Southeast and beyond.
Read More
Counting Butterflies

Butterflies are the oft-forgotten pollinators of Mississippi, but MAFES researchers are striving to change that. They have begun creating a massive inventory of the butterflies in the Sam D. Hamilton Noxubee National Wildlife Refuge. By making note of butterfly species, populations, and habitats, researchers can shape a fuller idea of the best management practices to keep the Refuge's butterflies thriving.
Read More
Disrupting the Green Bridge

A common concern among farmers first exploring the option of cover crops is the pests that use the cover crop as a "Green Bridge" onto the cash crop. Researchers at MAFES sought to determine the influence of winter annual cover crops and insect management strategies on surface and subsurface insect pests in soybeans. After conducting numerous variations of several management practices, they found neonicotinoid seed treatments and early termination of cover crops were sufficient in controlling pests and did not observe a "Green Bridge" phenomenon.
Read More
High-protein Insect Studied at MSU

MAFES scientists are studying the black soldier fly—an insect that can eat nearly anything, control microbes, live off water alone in the adult stage, and be a good source of protein for animal feed—as a potential solution to dealing with large amounts of waste while also generating a feed product. Harvested larvae can be dried and milled to create a high-protein meal for livestock, poultry and aquaculture consumption. Due to their high oil content, black soldier fly larvae may even be useful for biofuel production. They're not a known disease carrier, they don't bite or sting, and they're not a nuisance. They're a versatile species with huge potential.
Read More
Identifying Destructive Invaders

Exotic insect species enter the United States through multiple routes, such as on wood shipping pallets, plant materials, and imported fruits and vegetables. The U.S. government sets trade restrictions to help prevent the introduction of nonnative pests, and its inspectors work at all borders to search for and confiscate materials carrying these insects. Some hidden pests do make it past inspection and move into U.S. crops. Once established, these pests can damage crops and native plant species, ultimately causing severe economic damage. Quick identification of invasive species is crucial to stopping their spread. The Mississippi Entomological Museum was recently designated as the Eastern Region Identification Center for the USDA's Animal and Plant Health Inspection Service.
Invasive Beetle Threatens Avocados and Other Laurel Trees

The redbay ambrosia beetle carries a pathogen that causes laurel wilt disease and threatens all members of the laurel family in the U.S., which includes sassafras, redbay, and commercially important avocados. Dr. John Riggins, a forest entomologist with the Mississippi Agricultural and Forestry Experiment Station, is leading MSU efforts to battle the insect. Efforts to control the pest are focused on educational campaigns. Researchers continue to look at ways to address the problem. Part of their effort is simply learning all there is to know about the insect, its habitat, and its life cycle. The next step in the research is to find resistant trees and plant them in the environment.
Read More
MSU Research Takes the Bite Out of Mosquitoes

The Mississippi Department of Health (MDH) conducted pre- and post-grant surveys designed to gauge practices, knowledge and attitudes of local personnel in mosquito- control programs before and after disbursement of funds. Postdoctoral associate and veterinarian Kristine Edwards and associate Extension professor Jerome Goddard conducted research and workshops based on survey responses, where Edwards said simple tactics like surveying and trapping can save local governments time and effort.
Read More
MSU Researcher Finds New Grasshopper Species

Entomologists at MSU have been finding and naming new species for the last 100 years, and this includes several hundred species of beetles, moths, leafhoppers, wasps and other insects. JoVonn Hill and four others currently on the staff of the Mississippi Entomological Museum have described and named about 20 new species in recent years, including the never-before-identified grasshopper—now named Melanoplus ingrami—that Hill stumbled upon his path.
Read More
Targeting Pests

One of the most consistent challenges producers face is yield loss from insects. Over the years, strides have been made to develop cultivars, specifically of cotton and corn, that are naturally resistant to insects, thanks to a naturally occurring soil bacterium, Bacillus thuringiensis, or Bt, that is detrimental to common pests. Bt crops are created by inserting genes into the plant's genome, causing the plant to produce proteins toxic to certain insects. In modern agronomic insect pest management, Bt crops/technologies are some of the most effective tools. MAFES scientists at the Delta Research and Extension Center have been hard at work evaluating the field performance of these Bt crops including treatment thresholds for these crops when or if foliar insecticide applications are needed, and performance-issue events such as unexpected damage or insect survival. The study has shown some pests are becoming resistant to one of the detrimental proteins Bt crops produce, opening a new avenue of research to help producers mitigate insect pressure in the future.
Read More
The Buzz Surrounding Neonics

Neonicotinoids (also known as neonics) are systemic insecticides that are chemically similar to nicotine. A decline in managed pollinators over the last decade led researchers to investigate if neonicotinoids were a contributing factor. Researchers at Mississippi State University, the University of Arkansas and the University of Tennessee established collaborative research projects on the potential risks of neonicotinoids to managed pollinators. The scientists concluded that the risk to pollinators from neonicotinoids is so small; there essentially is no risk in the Midsouth.
Read More
Under Pressure

Insect pressure in the field has long been a problem for row crop producers. Additionally, insects can damage stored grain, which can be a huge nuisance for farmers and grain elevator operators. Dr. Fred Musser, a professor in the Department of Biochemistry, Molecular Biology, Entomology and Plant Pathology and MAFES scientist, and his team study corn earworms, soybean loopers, and tarnished plant bugs in row crops. He monitors insecticide efficacy and provides early warning when resistance occurs. Dr. Don Cook, MAFES researcher and associate research professor in the Department of Biochemistry, Molecular Biology, Entomology and Plant Pathology at the Delta Research and Extension Center, is monitoring insecticide efficacy in grain storage.
Read More
Vanquishing the Varroa

Biomedical, physiological, and agricultural experts across Mississippi are joining together to protect one very important creature: the bee. Worker bees everywhere are being plagued by insistent varroa mites, and MAFES researchers are bringing medical technology intended for humans into the hive to administer a more effective, longer-lasting method for fighting the varroa. Researchers hope the technology will provide a more effective way to eradicate the honeybee's biggest threat.
Read More
Plant Pathology
Corn Researchers Develop In-Field Aflatoxin Approach

Scientists at Mississippi State University's Delta Research and Extension Center are researching new ways to reduce aflatoxin in infected corn, a fungus that can reduce profits and hurt marketability. MSU researchers have partnered with researchers from the USDA to obtain strains of the fungus that do not produce aflatoxin in hopes of protecting much of the state's corn harvest.
Read More
Cotton Yields Increase With New Technology

Researchers at Mississippi State University have developed technology that uses reflected light to analyze the presence of certain nematodes in cotton fields so producers can increase profits. The data was used to generate a field map showing areas of low, medium and high nematode populations. From that, a prescription map for applying different amounts of nematicide was created. For the producers, the yields were higher, which increases profits. Plus, they saved money by applying only the amount of chemical required rather than blanketing their field with the amount needed to treat the highest population of nematodes found in their soil samples. The third benefit is to the environment, because site-specific applications reduce the amount of chemical used.
Read More
MSU Leads the Charge Against Soybean Taproot Decline

Soybean taproot decline (TRD) has been compromising crop yields in the Southeast for more than a decade, but researchers have now identified the fungus that causes this disease. MAFES scientists have been part of a cooperative regional effort to make the discovery. The average amount of loss caused by the fungus is about 18 percent of harvestable yield; however, soybean research pathologists conducted field trials during 2016 at the Delta R&E Center to assess best management practices. Possible strategies may include rotation, identification of the host range for this particular fungus, and screening commercially available fungicides to determine if anything currently on the market is efficacious against the fungus.
MSU Research Benefits Poinsettia Producers

MAFES researchers have found a way to use organic methods and fewer fungicides to successfully fight one of the most serious soil-borne diseases in poinsettia production. One of the strategies MAFES scientists are researching is integrating biofungicides. Biological agents are not conventional fungicides or chemicals, but organic methods of fighting fungi or other harmful microorganisms. The study found that a reduced rate of the conventional fungicide, when used with a biological agent, resulted in plants that didn't rot and had similar growth to the label rate of conventional fungicides. This is beneficial to growers because it reduces their impact on the environment. It also reduces the risk of the pathogens adapting to the fungicides and becoming resistant and may save producers money by reducing the amount of fungicide they use.
Read More
New Research to Aid State's Soybean Growers

The soybean cyst nematode, or SCN, is a small, plant-parasitic roundworm that attacks the roots of soybeans and leaves crop-rotation as the only defense for farmers. Researchers are in the process of developing new, nematode-resistant soybean varieties and are modifying current crop-rotation techniques in the meantime. They have developed a new test that uses a gene marker technique that can identify nematode races in a molecular laboratory in a matter of hour and allow farmers to choose varieties that are not susceptible to the particular nematodes in their fields.
Read More
Resist and Detect

While MAFES researchers have explored various methods for either removing the fungi-induced toxin, aflatoxin, from corn, or preventing it from forming in the first place, the answer may lie in bolstering the corn's ability to resist aflatoxin. Breakthroughs in gene sequencing technology have allowed scientists to identify exactly which genes carry resistance. They hope to then use CRISPR technology, which allows scientists to edit the genome of an organism to produce specific traits.
Read More
Root Cause

Taproot decline is the seventh-leading cause of yield loss for soybean producers across Mississippi, and Xylaria, the novel fungal pathogen behind the disease, has become a top research priority for a group of MAFES researchers. As the focus of an ongoing six-year study into the pathogen, these scientists are now striving to understand the soybean's immune response to Xylaria infection. While taproot decline is devastating to soybean producers, this study offers hope, as the immune response suggests that certain microbes in the soil could prevent infection. Fifteen of the more than 300 bacterial varieties tested for Xylaria resistance have shown promising results, and though the testing on which is best suited for large scale application continues, the researchers hope to one day have a microbial soil treatment to stop this disease.
Read More
Unlocking Mysteries

Dr. Maria Tomaso-Peterson, a research professor in biochemistry, molecular biology, plant pathology and entomology, recently classified Culvaria malina, a strain of fungus common in turfgrass, which had previously gone unclassified. In order to better understand the mystery fungus, Tomaso-Peterson set up greenhouse trials to examine which grasses were most susceptible. By identifying the pathogen and then classifying fungi according to genus-species, scientists are able to create a body of work, recording data such as the conditions it is found under, the symptoms it causes, and the biology behind it. It also allows them to get an idea of how prevalent it is in plants within a region, and what steps might be taken to prevent plant diseases.
Read More



